Industrial design, safety and fluent usability since 1901
Merivaara Group is a forerunner in intuitive healthcare technology and industrial design, with surgery room solutions being a key focus and growth area. Functionality and ease of use have been at the heart of the Merivaara design since 1901, when the company was founded. Today, Merivaara’s innovative products and solutions improve patient safety and increase quality and efficiency of surgery operations in leading international hospitals.
Made in Finland and investing in user-friendly design have served as the cornerstones of the company from the very beginning. This is also demonstrated by the Design from Finland mark awarded to Merivaara in 2021. The products and services that carry the mark are designed to be professional, user-friendly, and sustainable. Merivaara is part of the Finnish Lojer Group.
Merivaara products and solutions to surgery rooms
We offer to healthcare a wide range of high-quality products and solutions.
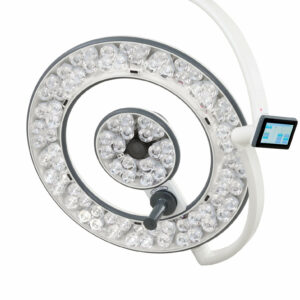
Merivaara’s Advanced Medical Lights Help to Speed up Post-Covid Medical Care
Merivaara has added new medical lights to its award-winning Q-Flow family. Lights are in high demand worldwide as medical professionals strive to shorten post-Covid waiting lists.
Read moreMeet us on Arab Health 2024 in Dubai
We have great pleasure to invite you to visit us at Arab Health 2024, which will be held in Dubai from 29. January - 1. February 2024.
Read moreSwedish orders worth of millions for Merivaara
The OpenOR operating room integration and AV system has been a success in Swedish hospitals. The Swedish subsidiary Merivaara AB has won more than 90% of the tenders in which...
Read more